LavaLAMP DNA Master Mix
Product Details
ShowBoth LavaLAMP DNA Master Mix kits contain: LavaLAMP DNA Master Mix, DNA Positive Control LAMP Primer Mix, and DNA Positive Control. The LavaLAMP DNA Master Mix with Dye also contains Green Fluorescent Dye for fluorescent detection of amplified DNA.
LavaLAMP DNA Master Mix
Key features
Show- Loop-mediated isothermal amplification (LAMP): Facilitates running amplification reactions in difficult test environments, enabling use outside of a high-tech laboratory.
- Master Mix Format: Simplifies reaction setup and minimises optimisation required to produce the best assay results.
- High Thermal Stability: Highly thermostable enzyme (up to 90 °C for 5 min) enables preheating of reactions containing purified target DNA which, depending on the target, may increase assay sensitivity and decrease time to results.
- Elevated LAMP Reaction Temperature (68-74 °C):Improves primer specificity and reduces background amplification depending on the DNA target.
- Freeze-dry Compatible: Enables generation of room temperature stable test kits through lyophilisation.
Product information
The LavaLAMP DNA Master Mix is intended to simplify development and optimisation of DNA LAMP (loop-mediated isothermal amplification) reactions. LAMP kits are commonly available as multi-component kits that require optimisation (e.g. MgSO4, betaine, enzyme as well as temperature, primer concentration, etc.). The LavaLAMP DNA Master Mix greatly simplifies reaction optimisation by limiting optimisation to target specific components/conditions such as LAMP primer design, target concentration and reaction temperature. Additionally, this master mix is heat stable at 90 °C for ≤5 minutes, which enables the addition of a reaction preheating step which may, depending on the target, increase assay sensitivity and decrease time to results.
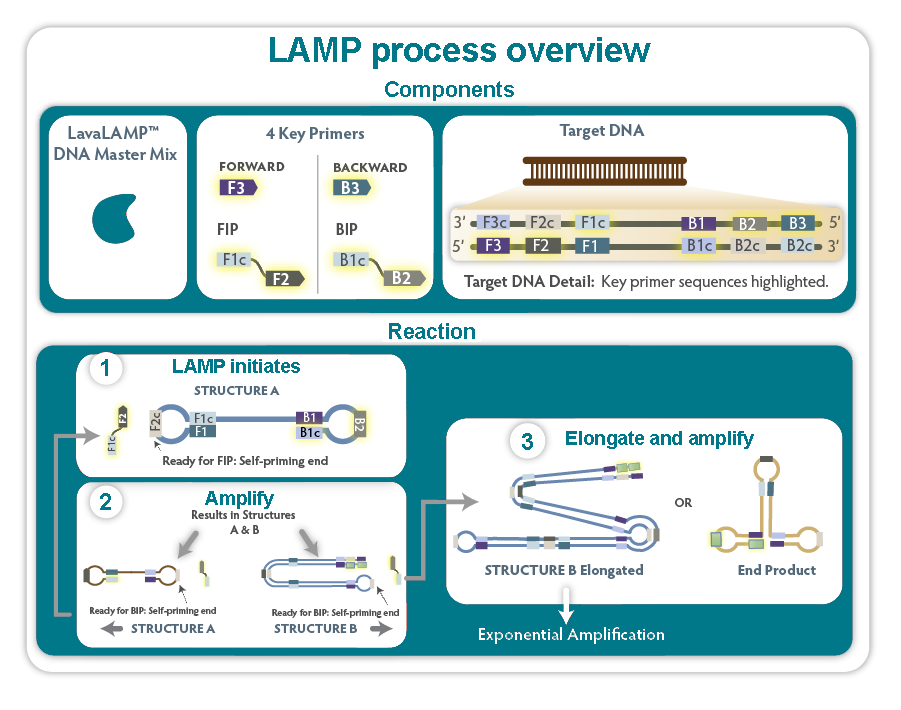
Figure 1. Simplified overview of the loop-mediated isothermal amplification (LAMP) process. For simplicity, 4/6 potential LAMP primers are illustrated. The inclusion of two additional primers, F-Loop; Forward Loop Primer and B-Loop; Backward Loop Primer, often significantly enhances amplification. For more information, please visit the Eiken website.
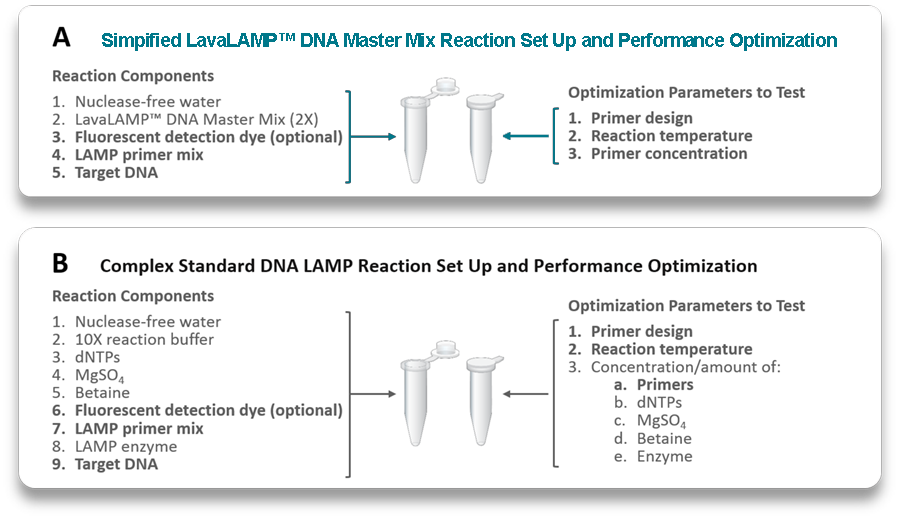
Figure 2. Illustration of the differences in LAMP reaction setup and potential optimisation parameters when using a master mix vs. a component kit format. Panel A illustrates the components added to a LAMP reaction (left) when using the LavaLAMP DNA Master Mix and the various parameters than can be optimised (right). Panel B also illustrates reaction setup (left) and potential optimisation parameters (right) when using a standard LAMP component kit.
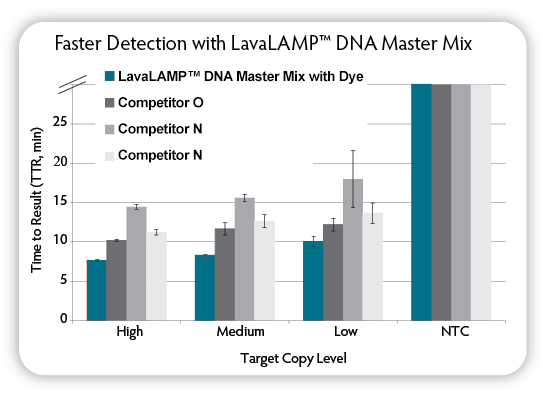
Figure 3. Loop-mediated isothermal amplification (LAMP) with real-time fluorescent detection of amplified products. LAMP reactions were set up using the indicated kits according to manufacturer’s recommendations. Target DNA ( C. difficile) at varying input amounts, tcdA target LAMP primers, and Green Fluorescent Dye (LavaLAMP Kit) were included in all reactions. Reactions were run on a CFX96 Thermal Cycler (Bio-Rad) at the following temperatures: LavaLAMP; 68 °C; other kits at the recommended 65 °C and fluorescence was measured over 60 minutes to determine the TTR. NTC = No Target Control.
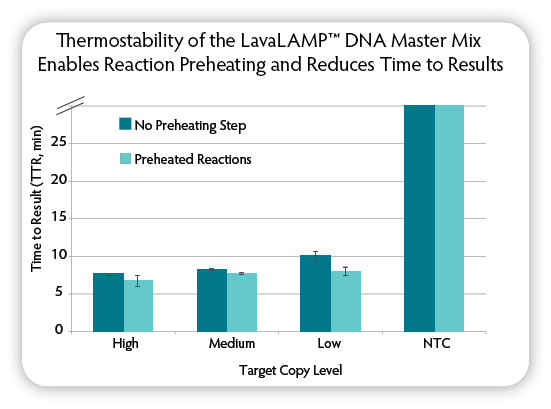
Figure 4. Testing the thermostability of the LavaLAMP DNA Master Mix. Two sets of triplicate LAMP reactions were set up using the indicated amounts of target DNA ( C. difficile), tcdA target LAMP primers, and Green Fluorescent Dye. One set of reactions was preheated to 90 °C for 5 minutes, and then all reactions were incubated for 60 minutes on a CFX96 Thermal Cycler (Bio-Rad) at 68 °C and fluorescence was measured during the course of the reactions to determine the TTR. NTC = No Target Control.
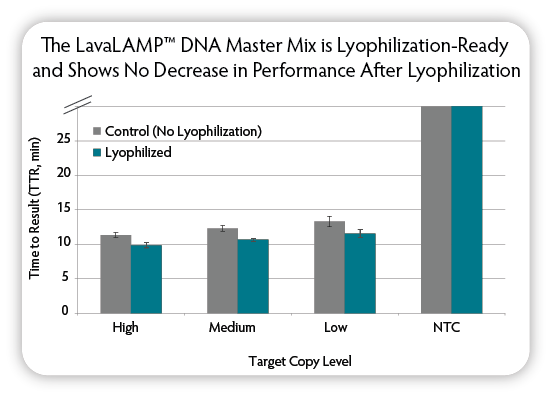
Figure 5. Testing the performance of lyophilised LavaLAMP DNA Master Mix. The LavaLAMP DNA Master Mix (12.5 µL) was aliquoted into tubes and lyophilised using a VirTis Wizard 2.0. After one day, each lyophilised tube was reconstituted in 12.5 µL water. Then six replicate LAMP reactions per condition were set up using the reconstituted lyophilised master mix and the standard LavaLAMP DNA Master Mix (Controls). The indicated amounts of target DNA ( S. aureus), clfA target LAMP primers, and Green Fluorescent Dye were added to the reactions. All reactions were incubated for 60 minutes in a CFX96 Thermal Cycler (Bio-Rad) at 68 °C and fluorescence was measured during the reactions to determine the TTR. NTC = No Target Control.
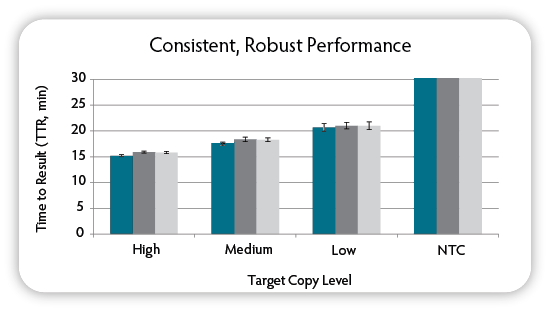
Figure 6. Analysis of day to day variability. On three different days, six replicate LAMP reactions were set up per condition using the indicated amounts of target DNA ( M13mp18), M13mp18 target LAMP primers, and Green Fluorescent Dye. On each day the reactions were run on a CFX96 Thermal Cycler (Bio-Rad), amplification was monitored in real-time, and then results were analysed to determine the TTR. NTC = No Target Control.
Licensing information: Lucigen is a fully licensed provider of LAMP reagents for research use. Patents WO 00/28082, WO 01/34790, and WO 01/77317 regarding the LAMP method are owned by the Eiken Chemical Co. Ltd. LavaLAMP™, OmniAmp® and Bst Polymerase, Exonuclease minus are sold by LGC, Biosearch Technologies under license for use in LAMP for research use only. The products may not be used for LAMP-based human or diagnostic purposes without obtaining a license from Eiken. US Patent 8093030 for LavaLAMP and OmniAmp is owned by LGC, Biosearch Technologies.
It is the sole responsibility of the buyer to ensure that use of the product does not infringe the patent rights of third parties. If the purchaser is not willing to accept these use limitations, LGC, Biosearch Technologies is willing to accept return of the product for a full refund.
SDS
-
30066 LavaLAMP DNA Master Mix
-
30067 LavaLAMP DNA Master Mix with Dye
-
EU IT 30066 1 LavaLAMP DNA Master Mix
-
EU IT 30067 1 LavaLAMP DNA Master Mix with Dye
Manuals and user guides
Product information sheets
Access support
Need some support with placing an order, setting up an account, or finding the right protocol?
Contact us